Advanced hemodynamic monitoring provides parameters beyond traditional vital signs (blood pressure [BP] and heart rate, e.g.). For patients with—or at risk of—circulatory failure, does the inclusion of additional hemodynamic data unnecessarily complicate diagnostic and therapeutic decision-making, particularly given the cognitive demands clinicians face in time-sensitive environments? Or does it actually simplify decision-making in ways that benefit patient outcomes?
The following common clinical scenarios illustrate how advanced hemodynamic monitoring may enhance decision-making efficiency, potentially improving patient management.
Advanced hemodynamic monitoring simplifies clinical decision-making by enabling the early detection of circulatory failure
In patients with circulatory failure, a reduction in cardiac output (CO) frequently precedes decreases in BP or changes in heart rate 1. Compensatory mechanisms, such as increased systemic vascular resistance, can temporarily maintain traditional vital signs within normal ranges despite declining CO. Consequently, a decline in CO serves as an early indicator of circulatory compromise—one that clinicians might overlook without advanced hemodynamic monitoring. This early detection could enable timely intervention, allowing clinicians to address circulatory insufficiency before it progresses to a more severe and often more complex state—driven by the interplay of multiple mechanisms (for example, vasoplegia exacerbating initial hypovolemia, or secondary myocardial ischemia)—and complicating therapeutic decision-making. Such proactive management may help mitigate adverse patient outcomes.
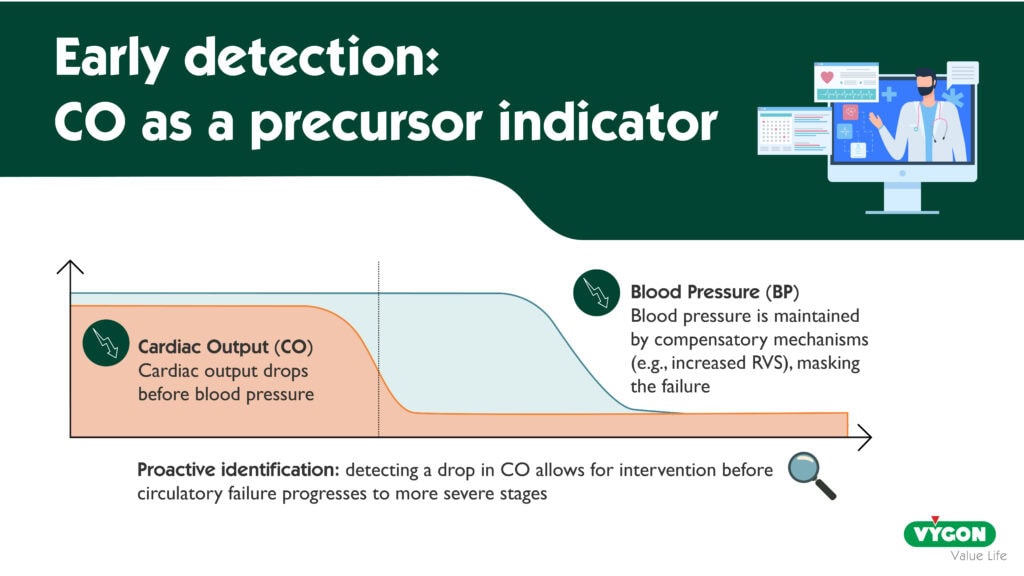
Advanced hemodynamic monitoring simplifies clinical decision-making by guiding the selection of appropriate interventions
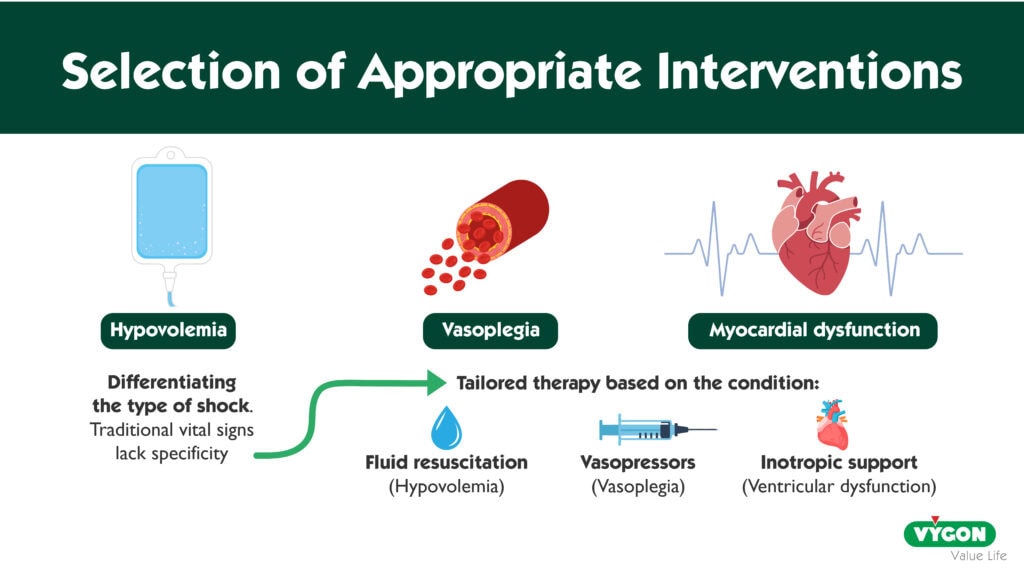
In both the operating room 2 and broader critical care settings 3, circulatory failure arises from diverse causes and mechanisms. Treatment must be tailored to the underlying pathology: for instance, fluid resuscitation is indicated for hypovolemia but may be ineffective—or even harmful—in cases of vasoplegia or left ventricular dysfunction.
Traditional vital signs alone often lack the specificity needed to distinguish the cause of circulatory compromise. A low BP with elevated heart rate could reflect excessive vasoplegia, hypovolemia, left ventricular systolic dysfunction, or another mechanism entirely. In such cases, monitoring CO and its trends could provide valuable diagnostic insights. Additionally, advanced hemodynamic monitoring offers further refinement through parameters such as myocardial performance metrics and indicators of vasoplegia.
By helping to identify the specific type of circulatory failure, advanced hemodynamic monitoring enables clinicians to select targeted therapies. Additionally, it helps monitor the response to treatment and the progression of shock 4.
Advanced hemodynamic monitoring simplifies clinical decision-making regarding the adjustment or discontinuation of initial treatment
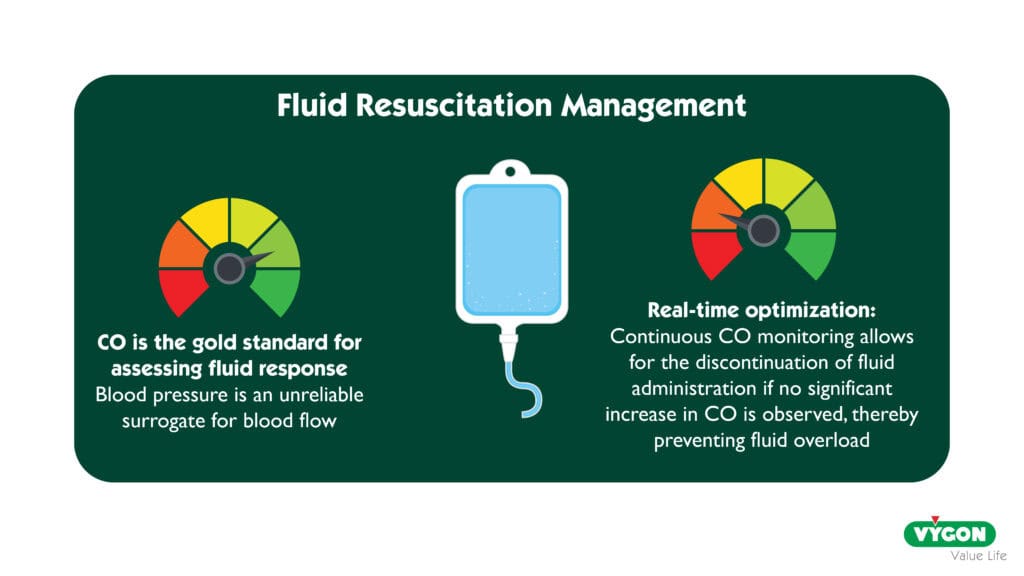
Let’s take, for example, fluid resuscitation. By increasing ventricular preload, fluid administration aims to enhance oxygen delivery through an increase in CO 5. However, like any therapeutic intervention, fluid administration carries both potential benefits—such as improving a compromised circulatory state—and risks, particularly if administered excessively or unnecessarily 6,7. Thus, fluid resuscitation should be carefully titrated, and consideration should be given to discontinuing it if it no longer produces a meaningful increase in CO. When CO measurements are unavailable, clinicians might instead monitor changes in BP to assess the response to fluid administration. Yet, fluid-induced BP changes are often an unreliable surrogate for improvements in CO 8,9. BP is influenced not only by CO but also by systemic vascular resistance 5, which can itself be affected by fluid administration. A lack of BP response might incorrectly suggest that fluid administration should be stopped, even if CO continues to improve—a potentially beneficial effect. Access to continuous, real-time CO measurements is therefore ideal for guiding fluid administration in a simple and physiologically relevant manner. The same principle applies to the titration of other treatments for circulatory failure, such as inotropic or vasopressor agents.
Advanced hemodynamic monitoring may also help inform other clinical decisions
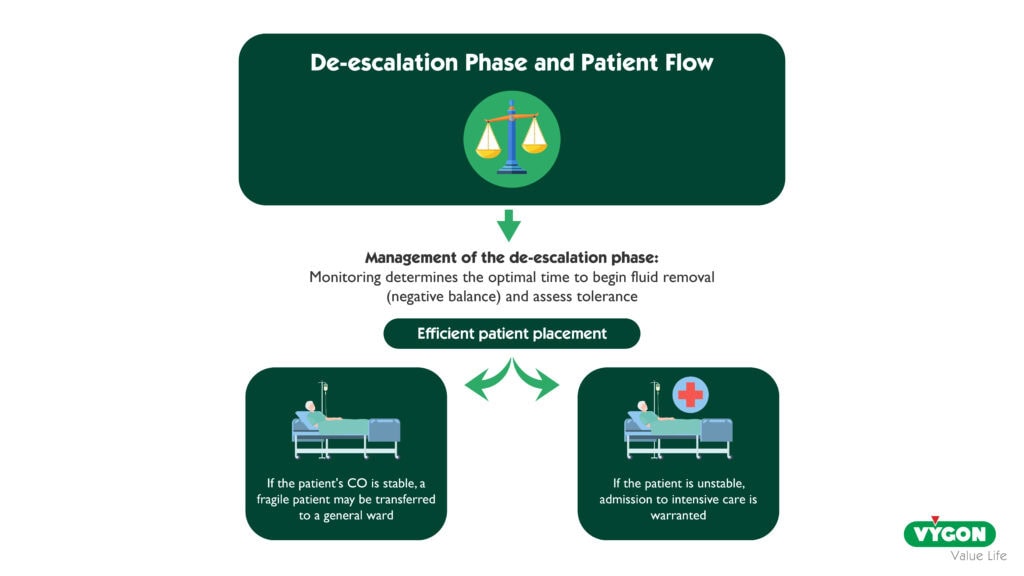
In the management of shock, after the resuscitation, optimization, and stabilization phases, the de-escalation phase follows 6,11. This involves achieving a negative fluid balance—using diuretic agents or, in some cases, extracorporeal fluid removal—to eliminate excess fluid from the tissues. Advanced monitoring could help determine the optimal timing for initiating fluid depletion and assessing its tolerability, both of which are important yet challenging aspects of managing a patient recovering from shock.
In the operating theatre, a common clinical dilemma is whether to transfer a frail postoperative patient to a standard ward or to an intermediate care unit. Accurate patient placement is essential: directing high-risk patients to units with enhanced monitoring could enable earlier identification and timely management of complications. However, inappropriate admission to intermediate care units can increase healthcare costs and staff workload, particularly given the often limited capacity of these units 12. Advanced hemodynamic monitoring—both during surgery and in the postoperative care unit—could help determine the appropriate postoperative destination. If CO remains stable, frail patients may be safely directed to a standard ward 12.
Advanced hemodynamic monitoring simplifies clinical decision-making by helping clinicians integrate and interpret parameters more effectively
To further simplify clinical decision-making, modern hemodynamic monitors are designed to be user-friendly, clearly displaying measured parameters and their trends. Additionally, these parameters can be integrated into visual decision-support tools, which may assist clinicians in synthesizing complex data and making informed choices 13.

IN CLINICAL PRACTICE
Among the many parameters provided by advanced hemodynamic monitoring—beyond traditional vital signs—CO is particularly pivotal 5,10. Incorporating CO into clinical reasoning does not significantly increase the clinician’s cognitive load. CO and its variations should therefore be the primary focus, in addition to traditional vital signs. Respiratory variations in BP or stroke volume can also inform the initial assessment, so long as their validity criteria are satisfied (e.g., absence of spontaneous breathing, cardiac arrhythmia, and adequate tidal volume).
As a second line of reasoning, the additional parameters provided by advanced hemodynamic monitoring further refine and optimize clinical decision-making.
THE BOTTOM LINE
In recent years, access to advanced hemodynamic monitoring has become significantly more straightforward. Less invasive methods are now available, eliminating the need for indicator injections or specialized sensors. Additionally, modern monitors have become increasingly user-friendly. Beyond the ease of access and use, the ability to measure parameters beyond traditional vital signs, especially CO, simplifies clinical decision-making and enhances its appropriateness.
The growing integration of artificial intelligence (AI) in next-generation monitors is expected to further streamline decision-making 14. By synthesizing the wealth of data provided by advanced hemodynamic monitoring, AI will assist clinicians—who will retain responsibility for the contextual interpretation of hemodynamic data.
REFERENCES
1. Ryan KL, Batchinsky A, McManus JG, Rickards CA, Convertino VA. Changes in pulse character and mental status are late responses to central hypovolemia. Prehosp Emerg Care 2008;12(2):192–8.
2. Joosten A, Chew MS, Futier E, et al. Intraoperative blood pressure management in noncardiac surgery: a narrative review based on current evidence. Intensive Care Med 2026;52(3):500–11.
3. Vincent J-L, De Backer D. Circulatory shock. N Engl J Med 2013;369(18):1726–34.
4. Monnet X, Messina A, Greco M, et al. ESICM guidelines on circulatory shock and hemodynamic monitoring 2025. Intensive Care Med 2025;51(11):1971–2012.
5. Mirus M, Saugel B, Spieth PM. Hemodynamic monitoring: basic principles in operation room and intensive care unit. J Clin Monit Comput 2026;40(2):375–89.
6. Malbrain MLNG, Langer T, Annane D, et al. Intravenous fluid therapy in the perioperative and critical care setting: Executive summary of the International Fluid Academy (IFA). Ann Intensive Care 2020;10(1):64.
7. Messmer AS, Zingg C, Müller M, Gerber JL, Schefold JC, Pfortmueller CA. Fluid Overload and Mortality in Adult Critical Care Patients-A Systematic Review and Meta-Analysis of Observational Studies. Crit Care Med 2020;48(12):1862–70.
8. Lakhal K, Ehrmann S, Perrotin D, Wolff M, Boulain T. Fluid challenge: tracking changes in cardiac output with blood pressure monitoring (invasive or non-invasive). Intensive Care Med 2013;39(11):1953–62.
9. Pierrakos C, Velissaris D, Scolletta S, Heenen S, De Backer D, Vincent J-L. Can changes in arterial pressure be used to detect changes in cardiac index during fluid challenge in patients with septic shock? Intensive Care Med 2012;38(3):422–8.
10. Fellahi J-L, Biais M, Abou-Arab O, et al. Perioperative hemodynamic optimization – Adults including obstetrics. Anaesth Crit Care Pain Med 2026;45(1):101662.
11. Ostermann M, Alshamsi F, Artigas Raventos A, et al. European Society of Intensive Care Medicine Clinical Practice Guideline on fluid therapy in adult critically ill patients: Part 3-fluid removal at de-escalation phase. Intensive Care Med 2025;51(10):1749–63.
12. Giacomelli E, Dorigo W, Romano SM, et al. The Use of Pressure Recording Analytical Method in Patients Undergoing Endovascular Repair for Abdominal Aortic Aneurysm: The Impact on Clinical Decisions for the Appropriate Postoperative Setting and Cost-effective Analysis. Acta Medica Acad 2024;53(1):10–23.
13. Michard F, Wong A, Kanoore Edul V. Visualizing hemodynamics: innovative graphical displays and imaging techniques in anesthesia and critical care. Crit Care 2025;29(1):3.
14. Michard F, Abou-Arab O. Hemodynamic phenotyping 4.0. Anaesth Crit Care Pain Med 2026;45(2):101647.