Every day in NICUs, clinicians face the challenge of supporting the fragile respiratory systems of preterm infants. Among the most critical decisions is determining how to deliver surfactant, a choice that directly influences outcomes such as mechanical ventilation exposure, lung injury, and respiratory health. With evolving evidence and increasing adoption of less invasive approaches, understanding the differences between LISA and INSURE has never been more essential.
This article is informed by a selection of recent scientific sources, including international guidelines, clinical studies, and systematic reviews comparing LISA and INSURE. It synthesizes evidence from multiple peer‑reviewed publications without detailing each individually, ensuring a balanced, reliable, and practice‑oriented perspective for neonatal healthcare professionals.
Definition of Respiratory Distress Syndrome (RDS)
Respiratory Distress Syndrome (RDS) remains one of the primary causes of morbidity and mortality among preterm infants. Its incidence rises sharply with decreasing gestational age, affecting approximately 90% of infants at 24 weeks and 80% at 28 weeks of gestation1. It results from alveolar collapse caused by pulmonary immaturity and surfactant deficiency with clinical signs emerging within minutes to hours after birth2. We therefore understand that surfactant therapy plays an essential role in RDS management making the understanding of the surfactant synthesis mechanism important.
Importance of Surfactant Therapy
Surfactant production begins around the 26th week of gestation and progressively increases until birth. Because this process is still incomplete in premature infants, a large majority are born with a surfactant deficiency.
Administering exogenous surfactant plays a critical role in restoring respiratory function by reducing alveolar surface tension, improving lung compliance, preventing atelectasis, and enhancing oxygenation3. The introduction of surfactant therapy has significantly increased survival rates, reduced the incidence of pneumothorax and bronchopulmonary dysplasia (BPD), and enabled shorter and less aggressive ventilation strategies. Early surfactant administration is particularly beneficial in very preterm infants1.
Emergence of Less Invasive Surfactant Administration Techniques
Traditional methods have long relied on intubation and mechanical ventilation, which, though effective for surfactant delivery, carry risks of volutrauma, barotrauma and BPD4.
To reduce complications related to invasive ventilation, two main strategies have emerged3. The INSURE (Intubate–Surfactant–Extubate) technique developed in the 1990s, followed by the LISA (Less Invasive Surfactant Administration) method in the 2000s.
Both approaches aim to minimize the use of mechanical ventilation during the early phases of RDS management5.
The INSURE Method: Definition and Practical Considerations
The INSURE technique consists of short-term intubation involving surfactant instillation through the endotracheal tube, and rapid extubation2. Because it follows a familiar airway‑management practice, INSURE remains relatively straightforward for clinical teams experienced in neonatal intubation, while also ensuring reliable surfactant delivery since correct endotracheal tube placement can be directly verified before administration3.
However, INSURE retains the inherent drawbacks of an intubation‑based approach. Certain hospitals’ protocol may require bag‑mask ventilation, which can increase the risk of ventilator‑induced lung injury2. In practice, clinicians more commonly use catheters or feeding tubes passed through the endotracheal tube, widespread misuse despite the fact that specific endotracheal tubes with lateral port are designed for surfactant administration. This leads to the interruption of the ventilation while the surfactant tube is inserted in the endotracheal tube for the drug administration. Moreover, the technique may also involve the use of Magill forceps to guide the endotracheal tube during its placement, which can further complicate the procedure.
The LISA Method: Definition and Practical Considerations
By maintaining non‑invasive ventilation throughout the procedure, LISA avoids endotracheal tube insertion and preserves the infant’s spontaneous, physiological breathing. This approach is fully aligned with current recommendations to favour non‑invasive ventilation during the transition after birth in preterm newborns. Dedicated devices designed for LISA enable surfactant administration without placing an endotracheal tube, and the procedure itself follows a simple sequence, visualising the glottis, inserting the thin catheter into the trachea, administering the surfactant, using air to gently push any residual surfactant left in the thin catheter, and then removing it. Evidence also suggests that LISA promotes a homogeneous distribution of surfactant within the lungs, thereby providing a good therapeutic effect2.
Although the technique may be considered less challenging than intubation, healthcare professionals still require performing laryngoscopy and proper training, and the method must be fully implemented within the unit to give teams the confidence to use it safely and effectively. With the LISA method, the frequency of neonatal intubation in routine practice decreases, making it more difficult to teach or to maintain this skill for clinical teams, a factor that can hinder full implementation3.
INSURE | LISA/MIST | ||
Advantages | Disadvantages | Advantages | Disadvantages |
Easier procedure, especially if you have experience intubating using an endotracheal tube. | If an ambu bag is used, lung damage can be induced. | Allows spontaneous breathing with CPAP support. | Requires more experience if performed with a soft thin tube and Magill forceps. |
Ensures administration in the airway as you can check ETT position. | If a single-lumen endotracheal tube is used, the ventilation is interrupted during the surfactant administration. | Less invasive surfactant administration technique: No intubation with endotracheal tube. | If complications occur, bag and mask ventilation may be needed. |
Early extubation can be challenging,especially in unstable infants. | Supported by the recommendation to use non-invasive ventilation (CPAP) during the transition at birth in preterm newborns. | ||
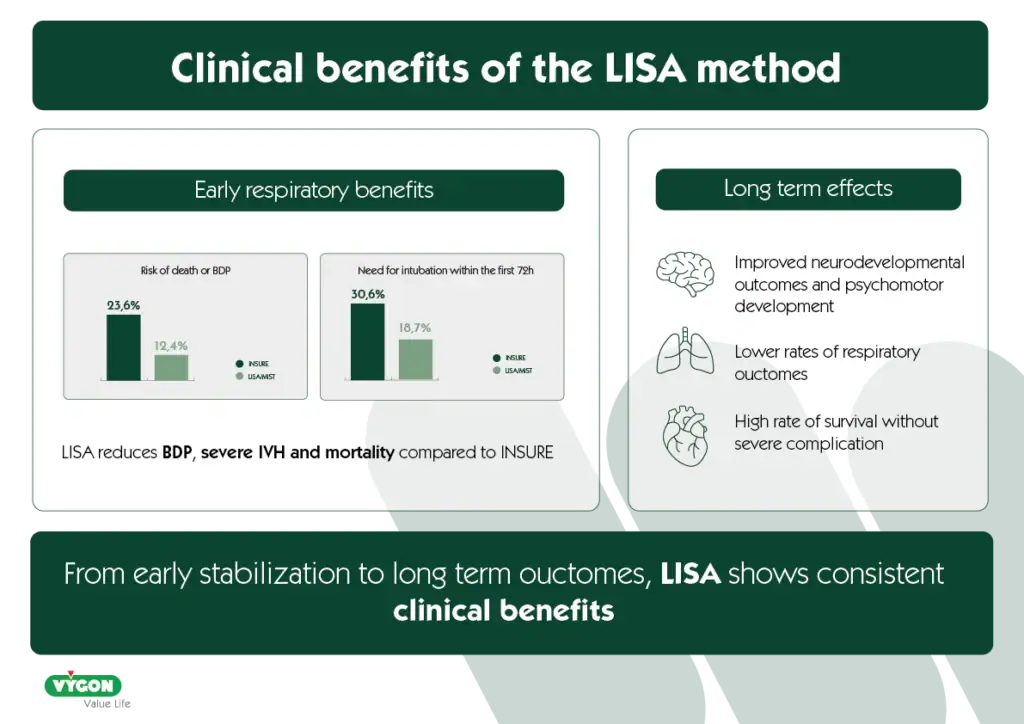
Clinical Results on the recent LISA technique
Recent evidence consistently demonstrates that the LISA method provides clear and consistent advantages, particularly among very preterm infants.
In infants born before 28 weeks, LISA is associated with a reduced need for mechanical ventilation during the first 72 hours of life6. This benefit is reinforced by systematic analysis, where intubation within the first 72 hours was required in only 18.7% of infants treated with LISA, compared with 30.6% of those managed with INSURE7. As well as improving early respiratory stability, LISA demonstrates improved morbidity and survival outcomes. European guidelines report lower rates of bronchopulmonary dysplasia (BPD), severe intraventricular hemorrhage (IVH), and mortality in direct comparisons with INSURE1. This trend is further illustrated by outcome of death or BPD, which was reduced to 12.4% under LISA, compared with 23.6% with INSURE7. Such improvements are aligned with the well‑established principle that limiting invasive ventilation helps prevent long‑term pulmonary complications3 .
Beyond the immediate benefits, several studies highlight long‑term effects of the LISA method, particularly in the most immature infants. Newborns treated with LISA show “lower rates of adverse respiratory outcomes during their first two years of life”8, as well as improved neurodevelopmental outcomes9 .
In addition, LISA has been associated with increased survival in extremely preterm infants10, with some studies showing its beneficial effects, such as reductions in severe IVH11. One report even highlights a high rate of survival without severe complications which was particularly remarkable as one in three infants were born before 24 weeks11. All together, these findings suggest that the advantages of LISA extend past immediate respiratory stabilization and may positively influence longer‑term developmental and clinical outcomes. In particular, infants born at 25 and 26 weeks have demonstrated better neurodevelopment, and those born at 23 and 24 weeks have shown improved psychomotor development two years after receiving LISA‑based surfactant therapy12.
Several studies also emphasize that LISA is feasible to integrate into routine NICU workflows and performs reliably even in centers without formalized training programs6. Its adoption continues to grow internationally, with NICUs incorporating the technique despite notable differences in devices used and catheter types4.
Conclusion: A Patient‑Centered Approach
While contemporary evidence consistently favors LISA for reducing mechanical ventilation and improving neonatal outcomes, the choice between LISA and INSURE must always remain individualized.
The techniques are not mutually exclusive, and both have well‑defined roles. Patient factors (gestational age, stability, airway anatomy, respiratory effort) must guide the choice. Clinical context, team expertise and available equipment are equally decisive.
Healthcare teams are encouraged to maintain proficiency in both LISA and INSURE while refining patient‑specific selection criteria. Reviewing local protocols, engaging in ongoing training, and fostering multidisciplinary discussions will help ensure that each baby receives the most appropriate and safest surfactant administration strategy. By staying aligned with emerging evidence and continuously evaluating clinical practice, NICU teams can optimise outcomes and contribute to advancing standards of care for preterm infants.
Bibliography
- European Consensus Guidelines on the Management of Respiratory DistressSyndrome: 2022 Update
- Araújo, V., da Silva, R. A., Pereira, M. O., Machado, C., & Raposo, F. (2024). Surfactant administration in preterm neonates with respiratory distress syndrome: Lisa versus insure. INTERNATIONAL JOURNAL OF PEDIATRICS, 6(2), 182-190.
- Sánchez L. Importance of surfactant in neonatal patients. Vyneodays Conference, november 2024, Valencia (Spain).
- Costa, S. D., Faria, S. S., Ferraz, C., & Azevedo, A. (2025). MIST/LISA in Portugal: National Survey of Clinical Practice. Journal of Paediatrics and Child Health.
- Kuitunen, I., & Räsänen, K. (2024). Less invasive surfactant administration compared to intubation, surfactant, rapid extubation method in preterm neonates: an umbrella review. Neonatology, 121(4), 485-493
- Heiring, C., Hedegaard, S. S., Carlsen, E. M., Kristensen, R., Breindahl, N., Schmidt, C., … & Aunsholt, L. (2025). Less Invasive Surfactant Administration Versus Intubate‐Surfactant‐Extubate: Associated With Reduced Mechanical Ventilation in Extremely Preterm Infants. Acta Paediatrica, 114(8), 1868-1876.
- Abdel-Latif ME, Davis PG, Wheeler KI, De Paoli AG, Dargaville PA. Surfactant therapy via thin catheter in preterm infants with or at risk of respiratory distress syndrome. Cochrane Database of Systematic Reviews 2021, Issue 5. Art. No.: CD011672. DOI: 10.1002/14651858.CD011672.pub2.
- Dargaville PA, Kamlin COF, Orsini F, Wang X, De Paoli AG, Kanmaz Kutman HG, Cetinkaya M, Kornhauser-Cerar L, Derrick M, Özkan H, Hulzebos CV, Schmölzer GM, Aiyappan A, Lemyre B, Kuo S, Rajadurai VS, O’Shea J, Biniwale M, Ramanathan R, Kushnir A, Bader D, Thomas MR, Chakraborty M, Buksh MJ, Bhatia R, Sullivan CL, Shinwell ES, Dyson A, Barker DP, Kugelman A, Donovan TJ, Goss KCW, Tauscher MK, Murthy V, Ali SKM, Clark HW, Soll RF, Johnson S, Cheong JLY, Carlin JB, Davis PG; OPTIMIST-A Trial Investigators. Two-Year Outcomes After Minimally Invasive Surfactant Therapy in Preterm Infants: Follow-Up of the OPTIMIST-A Randomized Clinical Trial. JAMA. 2023 Sep 19;330(11):1054-1063. doi: 10.1001/ jama.2023.15694. PMID: 37695601; PMCID: PMC10495923.
- Kakkilaya V, Gautham KS. Should less invasive surfactant administration (LISA) become routine practice in US neonatal units? Pediatr Res. 2023 Apr;93(5):1188-1198. doi: 10.1038/ s41390-022-02265-8. Epub 2022 Aug 19. PMID: 35986148; PMCID: PMC9389478.
- Härtel C, Herting E, Humberg A, Hanke K, Mehler K, Keller T, Mauer I, Frieauff E, Meyer S, Thome UH, Wieg C, Schmidtke S, Kribs A, Göpel W; German Neonatal Network. Association of Administration of Surfactant Using Less Invasive Methods With Outcomes in Extremely Preterm Infants Less Than 27 Weeks of Gestation. JAMA Netw Open. 2022 Aug 1;5(8):e2225810. doi: 10.1001/jamanetworkopen.2022.25810. Erratum in: JAMA Netw Open. 2023 Apr 3;6(4):e2312619. doi: 10.1001/ jamanetworkopen.2023.12619. PMID: 35943742; PMCID: PMC9364126.
- Klein R, Fastnacht L, Kribs A, Kuehne B, Mehler K. LISA Eligibility and LISA Success in Extremely Preterm Infants: A Single-Center Experience. Neonatology. 2024;121(4):530-535. doi: 10.1159/000537904. Epub 2024 Apr 10. PMID: 38599191; PMCID: PMC11318578.
- Mehler K, Broer A, Roll C, Göpel W, Wieg C, Jahn P, Teig N, Höhn T, Welzing L, Vochem M, Hoppenz M, Bührer C, Franklin J, Roth B, Herting E, Kribs A. Developmental outcome of extremely preterm infants is improved after less invasive surfactant application: Developmental outcome after LISA. Acta Paediatr. 2021 Mar;110(3):818-825. doi: 10.1111/apa.15565. Epub 2020 Sep 25. PMID: 32892376.






