Reducing Contamination Risk Through Better Practice and Better Device Design
Cleanliness in neonatal enteral feeding is not simply a procedural requirement; it is a fundamental safeguard for some of the most fragile patients in healthcare. A single lapse in hygiene can have severe consequences for premature and critically ill infants, whose immune systems are not yet equipped to defend against even small bacterial exposures.
In neonatal units, where expressed breast milk and formula are handled dozens of times per shift and enteral connectors are opened and closed repeatedly, maintaining rigorous cleanliness is both essential and challenging. Device design plays a crucial role: when components increase contamination risk, or require additional cleaning procedures, the burden on caregivers grows and so does the potential for harm.
Why Do Neonatal Units Face Elevated Contamination Risks?
- Immature Immune Defences
Preterm infants have limited antibody production, reduced skin integrity, and underdeveloped mucosal defences. Pathogens that would be harmless to an adult can rapidly escalate into serious infection in this population.
- Breast Milk is Biologically Active but Not Sterile
Human milk provides immunological benefits, but its nutrient-rich composition also makes it susceptible to bacterial growth if mishandled. Every transfer – from pump to bottle, bottle to syringe, syringe to tubing – creates opportunities for contamination.
- High Workflow Demands Increase Human Error Risk
Neonatal nurses routinely manage multiple complex tasks. As workloads intensify, routine cleaning steps or aseptic technique can become harder to sustain perfectly. Device systems that create more handling points naturally elevate contamination risk.
Cleanliness Is Not Just Technique – It’s a System
Even expert clinicians cannot fully compensate for equipment that inherently increases contamination risk. Cleanliness must be supported by devices designed to minimise touchpoints.
Key Contamination Touchpoints Include:
- Opening protective caps and connectors
- Transferring milk between containers
- Repeatedly connecting/disconnecting syringes
- Handling threaded components that can trap residue
- Managing tubing with residual milk inside
Every interface is an exposure point. In neonatal care, each exposure must be minimised.
ENFit® Connectors: Improved Safety, New Cleanliness Challenges
ENFit® connectors were introduced to prevent life‑threatening misconnections between enteral and parenteral lines- a major safety improvement that protects infants from wrong‑route errors.
However, ENFit® introduces its own cleanliness challenges due to the threaded design and fluid displacement mechanism.
Fluid Displacement Leaves Residue on the Connector Surface
When ENFit® connections are used, fluid displacement during the twisting motion causes small amounts of milk or enteral solution to be forced outward, where it adheres to:
- the edge of the male or female connector, and/or
- the threaded protective cap designed to prevent external contamination.
This residue is almost unavoidable, even with careful technique.
If not cleaned effectively, residue can:
- become a site for bacterial growth
- compromise asepsis at the next connection
- increase contamination risk directly at the feeding interface
For neonates with vulnerable GI and immune systems, this is a significant concern.
GEDSA’s Mandatory ENFit® Cleaning Protocol
In response to this known issue, GEDSA—the Global Enteral Device Supplier Association—issued a formal cleaning procedure specifically for ENFit® connectors.
This protocol is:
- mandatory before starting enteral feeding with an ENFit® connector
- required at least every 24 hours, or
- whenever the connector is visibly soiled
The cleaning process involves using sterile water and a small cleaning implement to remove trapped residue from the threads and internal surfaces of the connector.
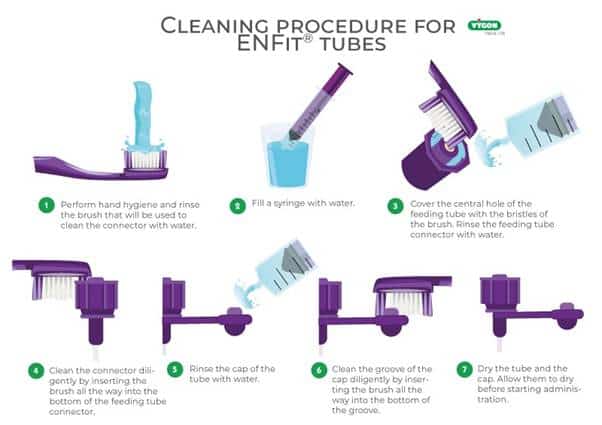
This protocol is essential to maintain cleanliness, but it also adds extra steps and time to the feeding workflow.
The Real-World Challenge: Cleanliness Under Pressure
While the GEDSA protocol provides a safety standard, its existence highlights a deeper issue:
When device design introduces contamination opportunities, clinicians must compensate with additional tasks.
In already high‑pressure environments, this means:
- increased cognitive load
- more steps during already time‑sensitive feeding prep
- reliance on perfect compliance to maintain safety
- additional stress during peak workloads or emergencies
It is a testament to nurses’ dedication that these procedures are followed as consistently as they are.
But the question remains:
Should device systems add steps, or should they eliminate them?
Cleanliness by Design: What Neonatal Units Truly Need
The ideal enteral feeding system for neonates would:
- minimise the number of open/close events
- use design geometry that does not trap milk residue
- eliminate unnecessary disconnections
- reduce the frequency and complexity of required cleaning
- protect connection points automatically
- enable safe handling even under time pressure
When cleanliness is engineered into the system, rather than added as an extra step, clinicians can focus on patient care instead of procedural compensation.
Next article:

Dose Accuracy: When Every Millilitre Matters
References
- Resources. (2016, 21 marzo). StayConnected by GEDSA. https://stayconnected.org/resources/
- Mehall, J. R., Kite, C. A., Saltzman, D. A., Wallett, T., Jackson, R. J., & Smith, S. D. (2002). Prospective study of the incidence and complications of bacterial contamination of enteral feeding in neonates. Journal of Pediatric Surgery, 37(8), 1177-1182.
- Mehall, J. R., Kite, C. A., Gilliam, C. H., Jackson, R. J., & Smith, S. D. (2002). Enteral feeding tubes are a reservoir for nosocomial antibiotic-resistant pathogens. Journal of Pediatric Surgery, 37(7), 1011-1012.
- Parker, L. A., Magalhães, M., Desorcy-Scherer, K., Torrez Lamberti, M., Lorca, G. L., & Neu, J. (2022). Neonatal feeding tube colonisation and the potential effect on infant health: a review. Frontiers in Nutrition, 9, 775014.
- Guenter, P., & Lyman, B. (2016). ENFit™ enteral nutrition connectors: benefits and challenges. Nutrition in Clinical Practice, 31(6), 769-772.
- Lyman, B. (2020). Randomized Controlled Trial Assessing the Effectiveness of Two Cleaning Regimens for ENFit™® Connectors. MedSurg Nursing, 29(6).

